RAISE
YOUR EXPECTATIONS
Explore our G-STRUT® metal framing channels and accessories, designed to complete your commercial and industrial projects with ease. From constructing new builds to retrofits, Gregory Strut's galvanized strut, pipe hangers, and rooftop supports deliver the flexibility and reliability you need to complete every job with confidence.
VIEW G-STRUT® CATALOG REQUEST A QUOTEINDUSTRIES WE SERVE
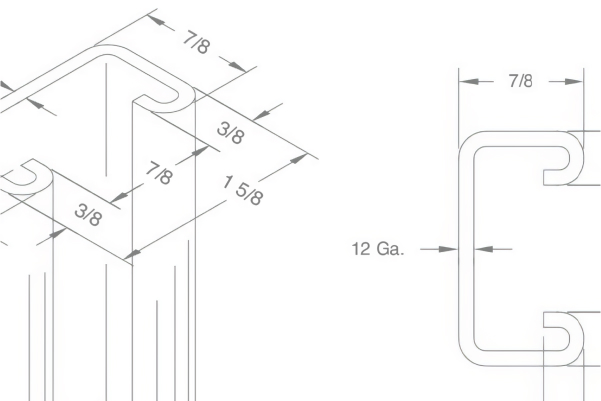
Specs & submittals
Looking to create a submittal sheet that is customized to your project?
VIEW SUBMITTAL SHEETS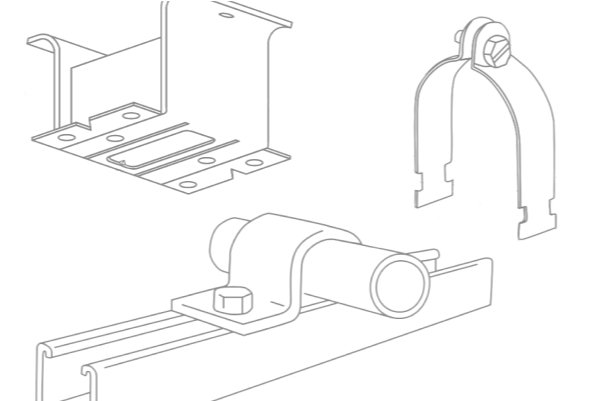
Part number lookup
Have a part number that belongs to another vendor? Use our interactive cross reference tool.
GET STARTEDGroup Affiliations




FIND A SALES REP
Contact one of our trusted sales reps to discuss your needs and request product details from our G-STRUT® catalog today.
GET IN TOUCH




































